Peroxide-forming compounds (PFCs) are chemicals that can react with oxygen in the air over time to form unstable peroxide crystals or liquids. These peroxides may be shock-sensitive, friction-sensitive, heat-sensitive, and capable of detonating during handling, opening, distillation, concentration, or disposal. Proper inspection, testing, labeling, and timely disposal are critical to maintaining a safe laboratory environment.
At University of Missouri, laboratories using peroxide-forming compounds must manage these materials proactively to prevent dangerous peroxide accumulation.
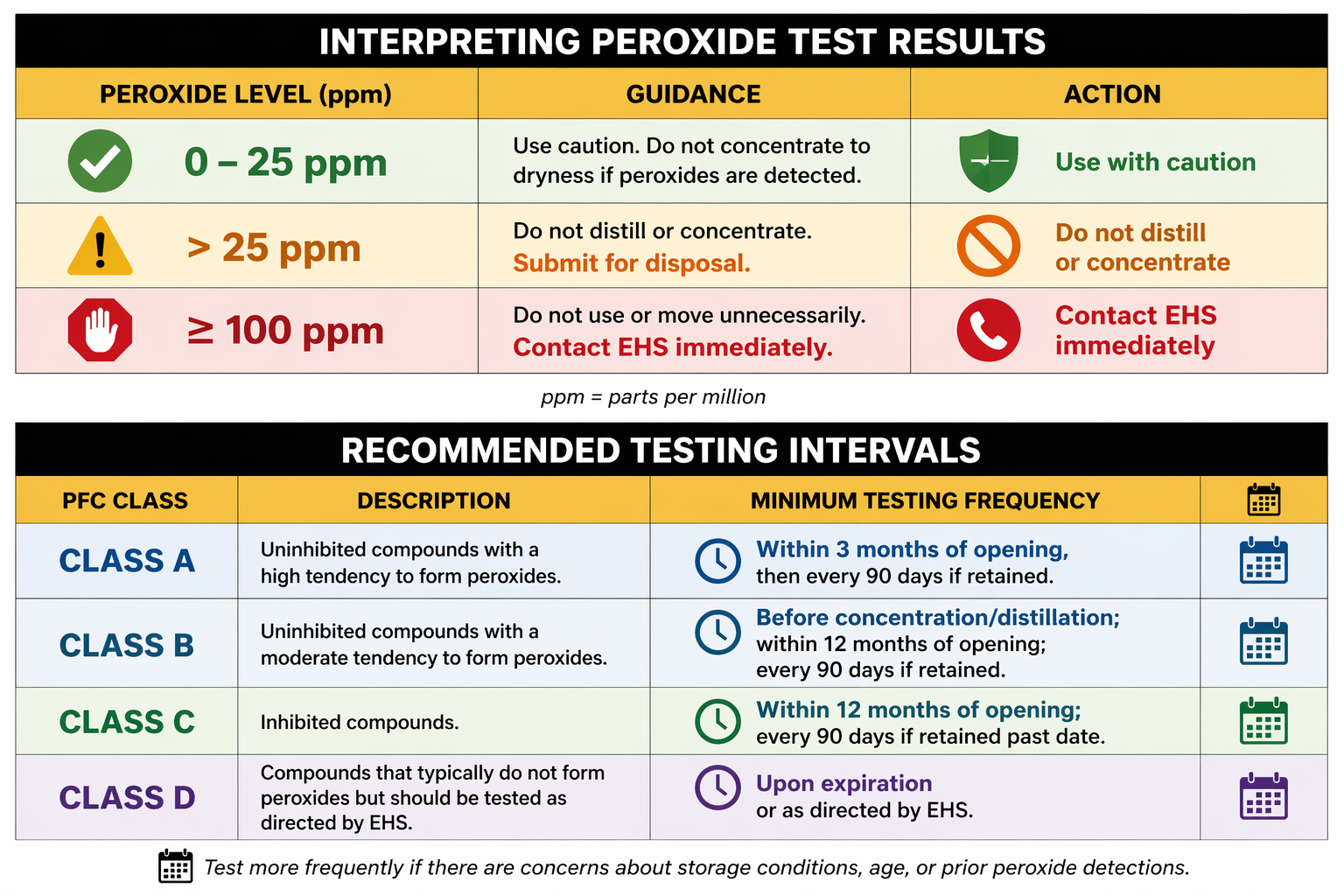
Testing is Important
Many peroxide-forming compounds appear normal even when hazardous peroxide concentrations are present. Routine testing helps identify materials that may no longer be safe to use.
Testing is especially important when:
- A container has been open for an extended period
- The manufacturer expiration date has passed
- The chemical will be distilled, evaporated, concentrated, or dried
- The material has unknown storage history
- There are concerns about improper storage conditions (heat, light, air exposure)
Peroxide formation can occur more rapidly in uninhibited materials. Exposure to oxygen, heat, ultraviolet light, and time all increase risk.

How to Test for Peroxides
Recommended Method: Commercial Peroxide Test Strips
Use peroxide test strips specifically designed for the chemical being tested.
Examples include:
- Organic solvent peroxide strips
- Hydroperoxide strips
- Aqueous peroxide strips (for water-based materials only)
Always follow the manufacturer’s instructions for the specific peroxide test strip being used. Environmental Health & Safety can provide a Peroxide Test Strip Starter Pack that includes 10 test strips along with reordering information to help laboratories begin routine peroxide monitoring. Contact EHS at (573) 882-7018 to request a starter pack or submit a supply request in EHSA.
General Procedure
- Put on required PPE (lab coat, gloves, eye protection).
- Work in a fume hood or safe ventilated area.
- Carefully transfer a small sample to a secondary container.
- Do not dip test strips directly into the original reagent bottle.
- Perform the test per strip instructions.
- Record results on the container label.
For many organic solvent strips, a drop of water may be needed after the solvent dries on the strip—follow the strip manufacturer directions carefully.
Safe Handling Reminders
- Purchase only the smallest practical quantity.
- Use older stock before opening new containers.
- Store tightly closed in a cool, dry place away from heat and light.
- Keep designated peroxide formers together in a managed storage area.
- Never return unused reagent to the original bottle.
- Dispose of expired material promptly