Biological Incident Response
Experiment Risk Assessment
The purpose of the risk assessment is to reduce potential hazards during acquisition, use, and storage of biohazardous materials. The Principal Investigator/Supervisor has responsibility for making the risk assessment and controlling the potential hazards associated with biohazardous materials. This should be done in close collaboration with the Institutional Biosafety Committee and MU Biosafety Officer. A written risk assessment must be conducted for each research protocol involving suspect biohazardous materials.
The FIRST STEP in the risk assessment process is to visualize the chain of infection within the research laboratory and/or animal facility (Refer to flow chart below).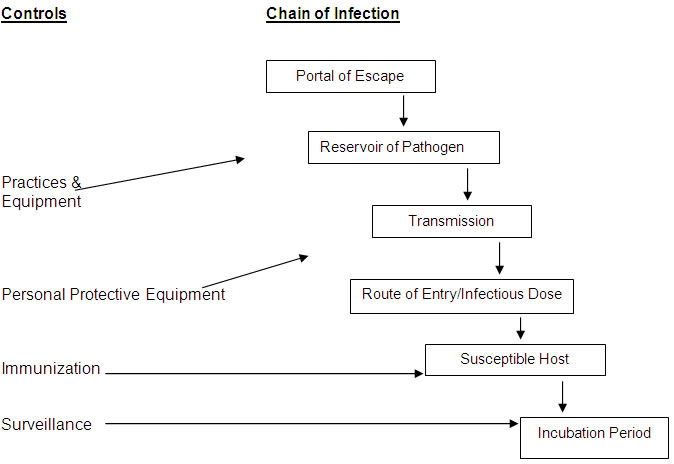
The SECOND STEP is to understand the risk factors associated with the specific biohazardous material to be used. Factors to be considered include, but are not limited to: agent identity and origin, virulence, pathogenicity, infectious dose, environmental stability, route of transmission and entry, communicability, operations, quantity, concentration, animal studies, availability of vaccine or treatment, and gene product effects such as toxicity, physiological activity and allergenicity. This information should be interpreted for its tendency to raise or lower the risk of laboratory-acquired infection. Refer to the information in the risk assessment resources.
The THIRD STEP is to identify proper classification of the biohazardous material such as Risk Group 1, 2 or 3.
The FOURTH STEP is to identify the proper controls such as biosafety containment (BSL1, 2 or 3/ABSL 1, 2 or 3) and safety procedures to interrupt the chain of infection within the laboratory and/or animal facility.
NOTE: Aerosol generation potential is a significant risk factor. Procedures with a potential for creating infectious aerosols or splashes include pipetting, centrifuging, grinding, blending, shaking, mixing, sonicating, opening containers of infectious materials, inoculating animals intranasally, harvesting infected tissues from animals or eggs, and necropsy of infected animals. (Per BMBL 6th edition, Section IV, page 40 & Section V, page 82).
